Surprising Facts About The Use Of Silver In Technology
It is common knowledge that Dmitri Mendeleev is generally credited for the publication of the periodic table, which is practically a tabular arrangement of all the chemical elements that we know so far. You even might be able to name most of these elements, but how they are created naturally or synthesized in laboratories is unknown to many people. Still, we will leave the genesis to the experts, but what we should take into consideration is how we utilize these classes of substances in everyday life.
Take, for instance, silver. Its light grayish color and lustrous traits may seem like a perfect component for making jewelry, tableware, and coins, but there is more to this precious metal than just designing shiny baubles. Its use in technology has tremendously expanded in recent years, so it is no wonder why there is such a high demand on the market. We won’t discuss its antecedent creation during the process of nucleosynthesis right after a supernova (we are all, in fact, made of stars), but we will see why silver’s unique technical proficiency has made it suitable for a wide range of applications.
Physical Traits
Silver is quite soft, ductile and easy to manipulate with due to its malleable traits, but it can be hardened by alloying. Its surface is effective at reflecting radiant energy (wavelengths longer than ~450 nm). As for its electrical conductivity, well, this is where silver reigns. The only downside is that it costs a lot, so copper is a more common solution. However, plates made particularly out of this element are employed to improve the electrical conductivity of particular parts and wires in radio-frequency engineering. Also, silver has the highest thermal conductivity (property of a material to conduct heat) of any metal, it is stable in both pure air and water, but it can tarnish if exposed long enough to air or water which contain ozone or hydrogen sulfide.

Use in Technology
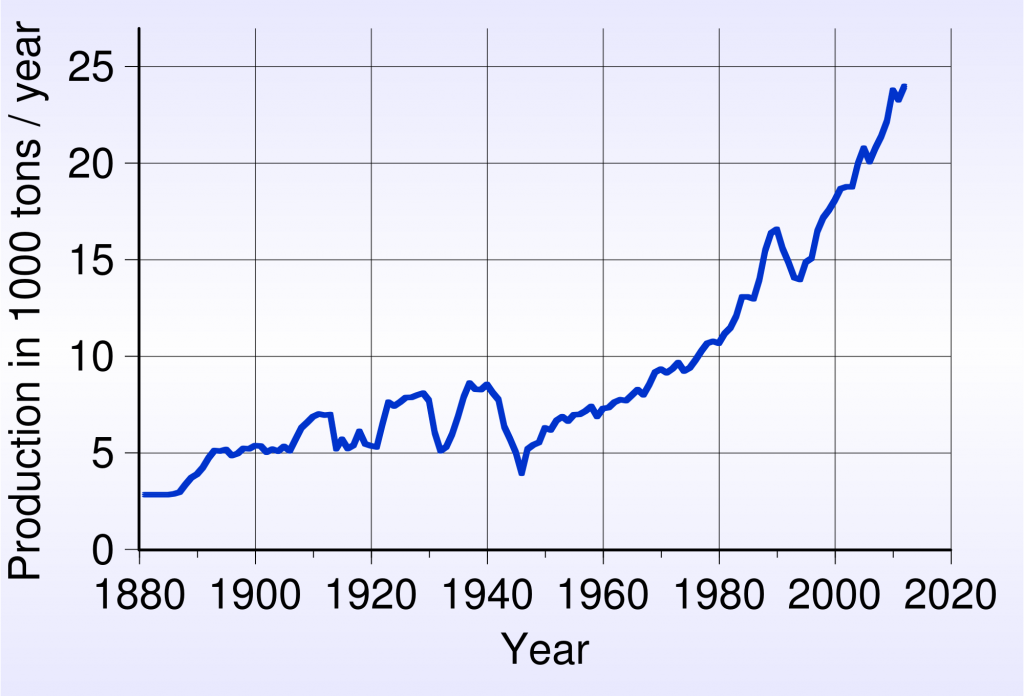
According to an article on oregonstate.edu, OSU scientists have solved that problem by using a micro reactor to create silver nanoparticles at room temperatures without any protective coating, and then immediately printing them onto almost any substrate with a continuous flow process. This gives silver bars providers and traders something to be happy about, because we are one step closer to synthetically producing this widely applicable precious metal in large quantities. Why is this a breakthrough?
Since silver is, without a doubt, one of the best electrical and thermal conductors, it is an ideal component for nearly all major fields of tech advancement and production, such as switches, conductive adhesives, contacts, ceramic capacitors, antifreeze, flexible plastics, hydraulics, etc. Even DVDs include traces of silver as a coating material because it is very useful for the optical data-storage media. Additionally, its natural antibacterial traits are incorporated into many medicinal applications and many lives have been saved in operating rooms because of it. The army loves to utilize silver with aluminum to create one of the world’s strongest alloys, plus it has high-duty bearings in aircraft engines.

Still, let’s not get ahead of ourselves with these rather unfamiliar uses of silver and switch to things we use on a daily basis. For instance, most of the electronic devices we possess – cell phones, plasma-display TVs, transparent displays, computers, laptops, etc. – contain this element. The reason it costs more than most metals is because it stays quite cool and it takes a lot of energy to produce enough heat for it to be manipulated. However, despite its complexity, manufacturers nowadays need silver for producing all manner of electronic wares, because it spells guaranteed quality.